how we work?
The quality systems EN ISO 13485:2016 and EN ISO 9001:2015 cover all activities such as designing, developing, manufacturing, purchasing in-vitro diagnostic medical devices and medical instruments.
more info
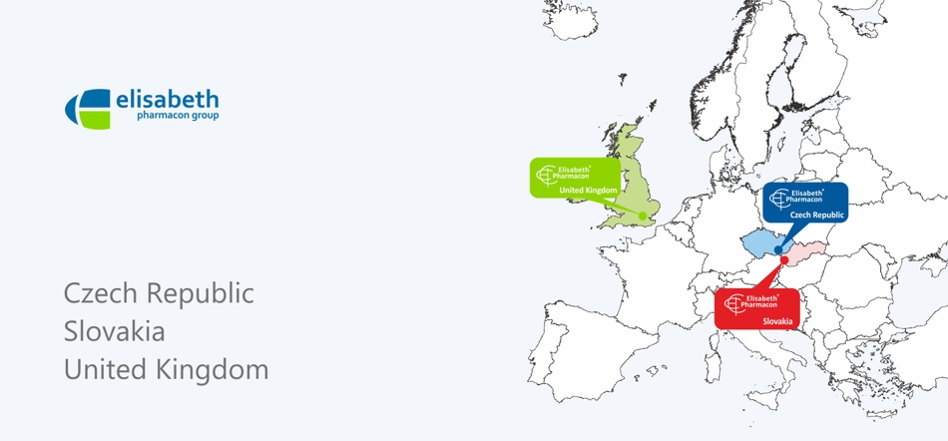
Worldwide solutions
Our world-class team of scientists is composed from university experts for DNA diagnostics, microbiology, virology, human genetics, animal and plant physiology, marketing, instrument constructions and robotics.
more info
Support center
All customers are important to us. Therefore, we provide professional technical support to help answer specialized questions of customers. Do not hesitate to contact our technical support.

Growth Strategies
ELISABETH PHARMACON GROUP had invested more than 3 million EUR in laboratory instruments and research. The research is continually supported by grant projects mostly from EU.